 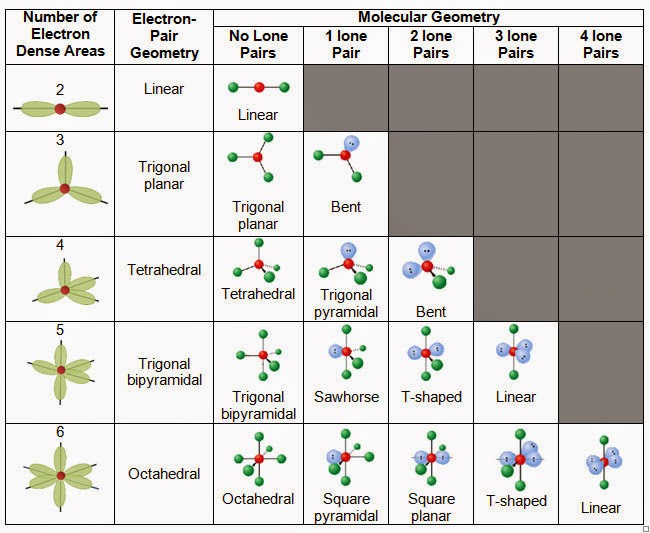
A single, double, or triple bond counts as one electron group. Count the number of electron groups or regions of electron density (lone pairs and bonds) around the central atom.The total valence electron in CH3F is 14. CH3F is polar due to its asymmetric shape. The molecular shape of CH3F is tetrahedral. Draw the Lewis structure of the molecule or polyatomic ion. CH3F is non-toxic gas which is also known as Fluoromethane or methyl fluoride.The Lewis electron-pair approach can be used to predict the number and types of bonds between the atoms in a substance, and it indicates which atoms have lone pairs of electrons. To predict whether a molecule has a dipole moment. The following procedure uses VSEPR theory to determine electron group geometry and molecular structures (molecular shape): To use the VSEPR model to predict molecular geometries. we can determine the bond angle to be about 105 degrees from our chart. This would make the electron geometry tetrahedral. Notice there are 4 attachments, or, electron groups surrounding oxygen. Predicting Electron Group Geometry and Molecular Structure The answer is the molecular geometry of water would be bent. 
The molecule has three atoms in a plane in equatorial positions and two atoms above and below the plane in axial positions. The molecular geometry of PCl 5 is trigonal bipyramidal, as shown in Figure R.2.3. The electron group geometries will be the same as the molecular structures when there are no lone electron pairs around the central atom, but they will be different when there are lone pairs present on the central atom. All electron groups are bonding pairs, so the structure is designated as AX 5. The structure that includes only the placement of the atoms in the molecule is called the molecular structure (or molecular shape). The three Cl atoms form an equilateral triangle. We differentiate between these two situations by naming the geometry that includes all electron pairs the electron group geometry. The geometry of BCl 3 is also given in Figure 7.2: it is trigonal planar, with all four atoms lying in the same plane, and all Cl B Cl bond angles equal to 120o. Molecular structure describes the location of the atoms alone, not including the lone pair electrons. Electron group geometries describe all regions where electrons are located, bonds as well as lone pairs. A The CHCl 3 molecule has four valence electrons around the central atom. It is important to note that electron group geometry around a central atom is not the same thing as its molecular structure. Together, the four sp 3 hybrid orbitals produce an approximately tetrahedral arrangement of electron pairs, which agrees with the molecular geometry predicted by the VSEPR model. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
